AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Angles in seesaw molecular geometry1/18/2024  
To know the total valence electrons of this compound, we need to know the valence electrons of both the atoms individually. The valence electrons that participate in forming bonds are called bonding pairs of electrons, whereas the electrons that do not participate or form any bonds are called nonbonding pairs of electrons or lone pairs.Īnd to draw the Lewis structure of SF4, we first need to know the total number of valence electrons in this molecule.Īs one can probably see, there is one sulfur atom in this compound and four fluorine atoms. The bonds formed between two atoms are depicted using lines, whereas the valence electrons not forming any bonds are shown by dots. Lewis structure is a pictorial representation of the bonds and valence electrons in the molecule. Hence, SF4 has a trigonal bipyramidal molecular geometry. The electrons follow this pattern of arrangement following the VSEPR rule to minimize the repulsion forces between the lone pairs of electrons to maximize the molecule’s stability. As there is one lone pair on the central atom, it repels the bonding pair of electrons, which tweaks the shape a little bit and makes it appear like a see-saw. Here two fluorine atoms forming bonds with the sulfur atom are on the equatorial positions, and the rest two are on the axial positions. Molecules having a molecular formula of AX4E have trigonal bipyramidal molecular geometry. 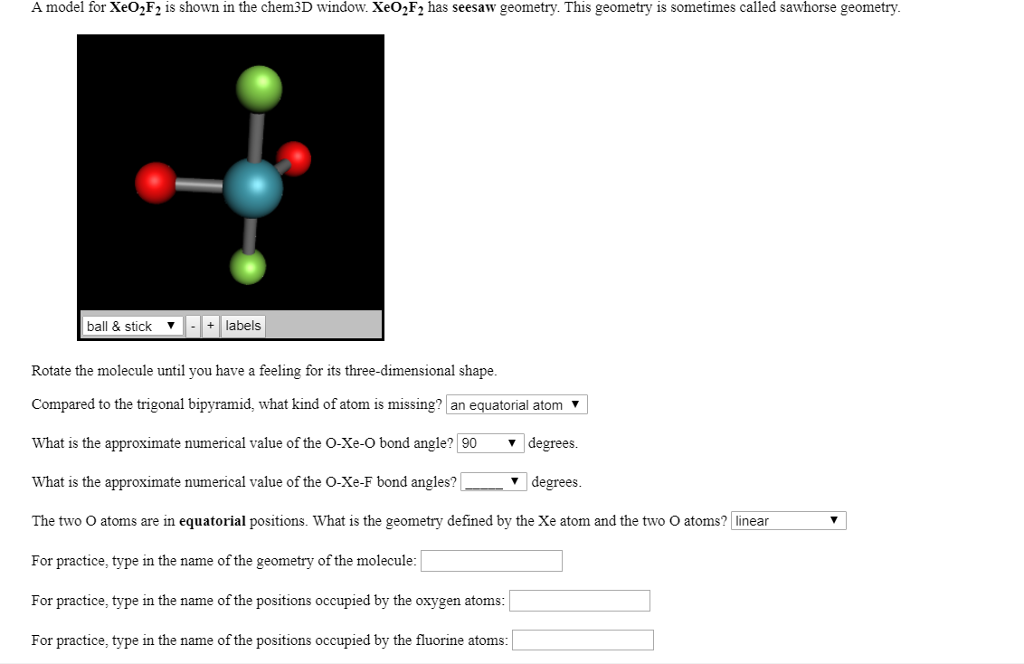
Here there is one sulfur atom and four fluorine atoms in the compound, which makes it similar to the molecular formula of AX4E. A molecular formula helps to know the exact number and type of atoms present in the given compound. It is easy to understand the molecular geometry of a given molecule by using the molecular formula or VSEPR model. SF4 Bond angles and shape SF4 Molecular Geometry. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
